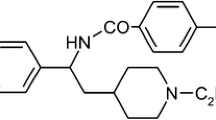
Abstract
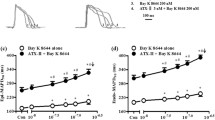
Rate-dependent repolarization (RDR) of action potential (AP) in cardiomyocyte plays a critical role in the genesis of arrhythmias and RDR in atrium has been linked with atrial fibrillation. However, detailed studies focusing on the role of RDR in rabbit atrium are scant. In this study, atrial cells were isolated from rabbit heart and rate-dependent property was explored in single atrial cell to elucidate the underlying mechanism. Our results indicated that rate-dependent prolongation was evident at the action potential duration at 20% (APD20) and 50% (APD50) repolarization but not at 90% repolarization (APD90) under control condition. Using transient outward potassium current (Ito) inhibitor 4-Aminopyridine (4-AP, 2 mM) effectively eliminated the changes in APD20 and APD50, and unmasked the rate-dependent reduction of APD90 which could be diminished by further adding L-type calcium current (ICaL) inhibitor nifedipine (30 μM). However, using the selective late sodium current (INaL) inhibitor GS-458967 (GS967, 1 μM) caused minimal effect on APD90 of atrial cells both in the absence and presence of 4-AP. In consistence with results from APs, Ito and ICaL displayed significant rate-dependent reduction because of their slow reactivation kinetics. In addition, the magnitude of INaL in rabbit atrium was so small that its rate-dependent changes were negligible. In conclusion, our study demonstrated that Ito and ICaL mediate RDR of AP in rabbit atrium, while minimal effect of INaL was seen.






Similar content being viewed by others
References
Attuel P, Childers R, Cauchemez B, Poveda J, Mugica J, Coumel P (1982) Failure in the rate adaptation of the atrial refractory period: its relationship to vulnerability. Int J Cardiol 2(2):179–197. https://doi.org/10.1016/0167-5273(82)90032-8
Belardinelli L, Liu G, Smith-Maxwell C, Wang WQ, El-Bizri N, Hirakawa R, Karpinski S, Li CH, Hu L, Li XJ, Crumb W, Wu L, Koltun D, Zablocki J, Yao L, Dhalla AK, Rajamani S, Shryock JC (2013) A novel, potent, and selective inhibitor of cardiac late sodium current suppresses experimental arrhythmias. J Pharmacol Exp Ther 344(1):23–32. https://doi.org/10.1124/jpet.112.198887
Boyett MR (1981) Effect of rate-dependent changes in the transient outward current on the action potential in sheep Purkinje fibres. J Physiol 319(1):23–41. https://doi.org/10.1113/jphysiol.1981.sp013889
Burashnikov A, Di Diego JM, Goodrow RJ Jr, Belardinelli L, Antzelevitch C (2015) Atria are more sensitive than ventricles to GS-458967-induced inhibition of late sodium current. J Cardiovasc Pharmacol Ther 20(5):501–508. https://doi.org/10.1177/1074248415570636
Christ T, Boknik P, Wohrl S, Wettwer E, Graf EM, Bosch RF, Knaut M, Schmitz W, Ravens U, Dobrev D (2004) L-type Ca2+ current downregulation in chronic human atrial fibrillation is associated with increased activity of protein phosphatases. Circulation 110(17):2651–2657. https://doi.org/10.1161/01.CIR.0000145659.80212.6A
Duan DY, Fermini B, Nattel S (1992) Sustained outward current observed after I(to1) inactivation in rabbit atrial myocytes is a novel Cl-current. Am J Phys 263:H1967–H1971
Fareh S, Villemaire C, Nattel S (1998) Importance of refractoriness heterogeneity in the enhanced vulnerability to atrial fibrillation induction caused by tachycardia-induced atrial electrical remodeling. Circulation 98(20):2202–2209. https://doi.org/10.1161/01.CIR.98.20.2202
Fermini B, Wang Z, Duan D, Nattel S (1992) Differences in rate dependence of transient outward current in rabbit and human atrium. Am J Phys 263:H1747–H1754
Giles WR, Imaizumi Y (1988) Comparison of potassium currents in rabbit atrial and ventricular cells. J Physiol 405(1):123–145. https://doi.org/10.1113/jphysiol.1988.sp017325
Grandi E, Pandit SV, Voigt N, Workman AJ, Dobrev D, Jalife J, Bers DM (2011) Human atrial action potential and Ca2+ model: sinus rhythm and chronic atrial fibrillation. Circ Res 109(9):1055–1066. https://doi.org/10.1161/CIRCRESAHA.111.253955
Guo D, Lian J, Liu T, Cox R, Margulies KB, Kowey PR, Yan GX (2011) Contribution of late sodium current (I(Na-L)) to rate adaptation of ventricular repolarization and reverse use-dependence of QT-prolonging agents. Heart Rhythm 8(5):762–769. https://doi.org/10.1016/j.hrthm.2010.12.026
Hou JW, Li W, Guo K, Chen XM, Chen YH, Li CY, Zhao BC, Zhao J, Wang H, Wang YP, Li YG (2016) Antiarrhythmic effects and potential mechanism of WenXin KeLi in cardiac Purkinje cells. Heart Rhythm 13(4):973–982. https://doi.org/10.1016/j.hrthm.2015.12.023
Jurkiewicz NK, Sanguinetti MC (1993) Rate-dependent prolongation of cardiac action potentials by a methanesulfonanilide class III antiarrhythmic agent. Specific block of rapidly activating delayed rectifier K+ current by dofetilide. Circ Res 72(1):75–83. https://doi.org/10.1161/01.RES.72.1.75
Lai LP, MJ S, Lin JL, Lin FY, Tsai CH, Chen YS, Huang SK, Tseng YZ, Lien WP (1999) Down-regulation of L-type calcium channel and sarcoplasmic reticular Ca(2+)-ATPase mRNA in human atrial fibrillation without significant change in the mRNA of ryanodine receptor, calsequestrin and phospholamban: an insight into the mechanism of atrial electrical remodeling. J Am Coll Cardiol 33(5):1231–1237
Li GR, Nattel S (1997) Properties of human atrial ICa at physiological temperatures and relevance to action potential. Am J Phys 272:H227–H235
Li GR, Yang B, Feng J, Bosch RF, Carrier M, Nattel S (1999) Transmembrane ICa contributes to rate-dependent changes of action potentials in human ventricular myocytes. Am J Phys 276:H98–H106
Li W, Yu Y, Hou JW, Zhou ZW, Guo K, Zhang PP, Wang ZQ, Yan JH, Sun J, Zhou Q, Wang YP, Li YG (2017) Larger rate dependence of late sodium current in cardiac Purkinje cells: a potential link to arrhythmogenesis. Heart Rhythm 14(3):422–431. https://doi.org/10.1016/j.hrthm.2016.11.036
Lindblad DS, Murphey CR, Clark JW, Giles WR (1996) A model of the action potential and underlying membrane currents in a rabbit atrial cell. Am J Phys 271:H1666–H1696
Luo A, Ma J, Song Y, Qian C, Wu Y, Zhang P, Wang L, Fu C, Cao Z, Shryock JC (2014) Larger late sodium current density as well as greater sensitivities to ATX II and ranolazine in rabbit left atrial than left ventricular myocytes. Am J Physiol Heart Circ Physiol 306(3):H455–H461. https://doi.org/10.1152/ajpheart.00727.2013
Moreno JD, Clancy CE (2012) Pathophysiology of the cardiac late Na current and its potential as a drug target. J Mol Cell Cardiol 52(3):608–619. https://doi.org/10.1016/j.yjmcc.2011.12.003
Nattel S (1999) Atrial electrophysiological remodeling caused by rapid atrial activation: underlying mechanisms and clinical relevance to atrial fibrillation. Cardiovasc Res 42(2):298–308. https://doi.org/10.1016/S0008-6363(99)00022-X
Poulet C, Wettwer E, Grunnet M, Jespersen T, Fabritz L, Matschke K, Knaut M, Ravens U (2015) Late sodium current in human atrial cardiomyocytes from patients in sinus rhythm and atrial fibrillation. PLoS One 10(6):e0131432. https://doi.org/10.1371/journal.pone.0131432
Qi D, Yang Z, Robinson VM, Li J, Gao C, Guo D, Kowey PR, Yan GX (2015) Heterogeneous distribution of INa-L determines interregional differences in rate adaptation of repolarization. Heart Rhythm 12(6):1295–1303. https://doi.org/10.1016/j.hrthm.2015.02.013
Ramirez RJ, Nattel S, Courtemanche M (2000) Mathematical analysis of canine atrial action potentials: rate, regional factors, and electrical remodeling. Am J Physiol Heart Circ Physiol 279(4):H1767–H1785
Shimizu W, Antzelevitch C (1997) Sodium channel block with mexiletine is effective in reducing dispersion of repolarization and preventing torsade des pointes in LQT2 and LQT3 models of the long-QT syndrome. Circulation 96(6):2038–2047. https://doi.org/10.1161/01.CIR.96.6.2038
Sossalla S, Kallmeyer B, Wagner S, Mazur M, Maurer U, Toischer K, Schmitto JD, Seipelt R, Schondube FA, Hasenfuss G, Belardinelli L, Maier LS (2010) Altered Na(+) currents in atrial fibrillation effects of ranolazine on arrhythmias and contractility in human atrial myocardium. J Am Coll Cardiol 55(21):2330–2342. https://doi.org/10.1016/j.jacc.2009.12.055
Van Wagoner DR, Pond AL, Lamorgese M, Rossie SS, McCarthy PM, Nerbonne JM (1999) Atrial L-type Ca2+ currents and human atrial fibrillation. Circ Res 85(5):428–436. https://doi.org/10.1161/01.RES.85.5.428
Wang Z, Feng J, Shi H, Pond A, Nerbonne JM, Nattel S (1999) Potential molecular basis of different physiological properties of the transient outward K+ current in rabbit and human atrial myocytes. Circ Res 84(5):551–561. https://doi.org/10.1161/01.RES.84.5.551
Workman AJ, Kane KA, Rankin AC (2001) The contribution of ionic currents to changes in refractoriness of human atrial myocytes associated with chronic atrial fibrillation. Cardiovasc Res 52(2):226–235. https://doi.org/10.1016/S0008-6363(01)00380-7
Workman AJ, Marshall GE, Rankin AC, Smith GL, Dempster J (2012) Transient outward K+ current reduction prolongs action potentials and promotes afterdepolarisations: a dynamic-clamp study in human and rabbit cardiac atrial myocytes. J Physiol 590(17):4289–4305. https://doi.org/10.1113/jphysiol.2012.235986
Wu L, Ma J, Li H, Wang C, Grandi E, Zhang P, Luo A, Bers DM, Shryock JC, Belardinelli L (2011) Late sodium current contributes to the reverse rate-dependent effect of IKr inhibition on ventricular repolarization. Circulation 123(16):1713–1720. https://doi.org/10.1161/CIRCULATIONAHA.110.000661
Yue L, Feng J, Gaspo R, Li GR, Wang Z, Nattel S (1997) Ionic remodeling underlying action potential changes in a canine model of atrial fibrillation. Circ Res 81(4):512–525. https://doi.org/10.1161/01.RES.81.4.512
Yue L, Melnyk P, Gaspo R, Wang Z, Nattel S (1999) Molecular mechanisms underlying ionic remodeling in a dog model of atrial fibrillation. Circ Res 84(7):776–784. https://doi.org/10.1161/01.RES.84.7.776
Funding
This work was supported by the State Key Program of National Natural Science Foundation of China (No. 81530015 to Yi-Gang Li), National Natural Science Foundation of China grant (No. 81270258 to Yi-Gang Li, Nos. 81370257, 81670414 to Yue-Peng Wang). and Shanghai City Committee of Science and Technology Research Projects (Nos. 12411951900, 13140903801, and 14441902502 to Yi-Gang Li).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Hou, JW., Li, W., Fei, YD. et al. ICaL and Ito mediate rate-dependent repolarization in rabbit atrial myocytes. J Physiol Biochem 74, 57–67 (2018). https://doi.org/10.1007/s13105-017-0603-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13105-017-0603-z